UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM
Current Report Pursuant
to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Date of Report (Date of earliest event Reported):
(Exact Name of Registrant as Specified in its Charter)
| (State or Other Jurisdiction of | (Commission File Number) | (I.R.S. Employer Identification Number) | ||
| Incorporation) |
(
(Addresses, including zip code, and telephone number, including area code, of principal executive offices)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class: | Trading Symbol(s) | Name of each exchange on which registered: | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging
growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 7.01. Regulation FD Disclosure.
The information in this Current Report (including Exhibit 99.1) is being furnished and shall not be deemed “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that Section. The information in this Current Report (including Exhibit 99.1) shall not be incorporated by reference into any registration statement or other document pursuant to the Securities Act of 1933, as amended, except as shall be expressly set forth by specific reference in such filing.
January 13-16, members of the Theravance Biopharma, Inc. management team will be conducting one-on-one meetings with analysts and investors and making a conference presentation in San Francisco, CA using a corporate slide presentation which is being furnished pursuant to Regulation FD as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated herein by reference.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits.
| 99.1 | Theravance Biopharma Investor Presentation dated January 2020 |
| 104 | Cover Page Interactive Data File (cover page XBRL tags embedded within the Inline XBRL document) |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| THERAVANCE BIOPHARMA, INC. | ||
| Date: January 13, 2020 | By: | /s/ Andrew Hindman |
| Andrew Hindman | ||
| Senior Vice President and Chief Financial Officer | ||
Exhibit 99.1

Corporate Presentation January 2020 THERAVANCE ® , the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE® are registered trademarks of the Theravance Biopharma group of companies. All third party trademarks used herein are the property of their respective owners. © 2020 Theravance Biopharma. All rights reserved.

Forward looking statements 2 Under the safe harbor provisions of the U . S . Private Securities Litigation Reform Act of 1995 , the company cautions investors that any forward - looking statements or projections made by the company are subject to risks and uncertainties that may cause actual results to differ materially from the forward - looking statements or projections . Examples of forward - looking statements in this presentation may include the Company’s strategies, plans and objectives, the Company’s regulatory strategies and timing of clinical studies (including the data therefrom), the potential characteristics, benefits and mechanisms of action of the Company’s product and product candidates, the potential that the Company’s research programs will progress product candidates into the clinic, the Company’s expectations for product candidates through development, potential regulatory approval and commercialization (including their differentiation from other products or potential products), product sales or profit share revenue and the Company’s expectations for its 2019 operating loss, excluding share - based compensation . The company’s forward - looking statements are based on the estimates and assumptions of management as of the date of this presentation and are subject to risks and uncertainties that may cause the actual results to be materially different than those projected, such as risks related to potential future disagreements with Innoviva, Inc . and TRC LLC, the uncertainty of arbitration and litigation and the possibility that an arbitration award or litigation result could be adverse to the Company, delays or difficulties in commencing, enrolling or completing clinical studies, the potential that results from clinical or non - clinical studies indicate the Company’s compounds or product candidates are unsafe or ineffective, risks that product candidates do not obtain approval from regulatory authorities, the feasibility of undertaking future clinical trials for our product candidates based on policies and feedback from regulatory authorities, dependence on third parties to conduct clinical studies, delays or failure to achieve and maintain regulatory approvals for product candidates, risks of collaborating with or relying on third parties to discover, develop, manufacture and commercialize products, and risks associated with establishing and maintaining sales, marketing and distribution capabilities with appropriate technical expertise and supporting infrastructure . Other risks affecting the company are described under the heading “Risk Factors” and elsewhere in the company’s Form 10 - Q filed with the Securities and Exchange Commission (SEC) on November 8 , 2019 , and other periodic reports filed with the SEC .

Strategic objective 3 Transform the treatment of serious diseases through the discovery, development, and commercialization of organ - selective medicines designed to maximize patient benefit while minimizing patient risk

Creating transformational value for stakeholders 1. TBPH holds 85% economic interest in upward - tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agreement over the next four fiscal quarters). 75% of royalties received pledged to service PhaRMA SM notes, 25% of royalties received retained by TBPH. All statements concerning TRELGY ELLIPTA based on publicly available information. 4 Innovative and productive research engine feeding pipeline of organ - selective assets Proven development expertise and established commercial infrastructure Strategic partnerships complement internal capabilities and balance technical, execution and financial risks Strong capital position augmented by TRELEGY ELLIPTA 1 royalties and YUPELRI ® launch Multiple milestones and value driving catalysts in 2020 and beyond

Program Indication Research Phase 1 Phase 2 Phase 3 Filed Marketed Collaborator Organ - selective YUPELRI® (revefenacin) LAMA COPD TD - 1473 GI JAKi UC CD TD - 8236 Inhaled JAKi Inflammatory lung diseases Wholly - owned TD - 5202 Irreversible JAK3i Inflammatory intestinal diseases New programs Multiple Wholly - owned Ampreloxetine (TD - 9855) NRI S ymptomatic neurogenic orthostatic hypotension Wholly - owned Program Indication Research Phase 1 Phase 2 Phase 3 Filed Marketed Rights Economic Interests TRELEGY ELLIPTA FF/UMEC/VI COPD GSK & Innoviva, Inc. Asthma Skin - selective JAKi Dermatological diseases Key programs supported by proven development and commercial expertise 1. TBPH holds 85% economic interest in upward - tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agreement over the next four fiscal quarters). 75% of royalties received pledged to service PhaRMA SM notes, 25% of royalties received retained by TBPH. All statements concerning TRELGY ELLIPTA based on publicly available information. FF/UMEC/VI: fluticasone furoate/ umeclidinium /vilanterol; comprised of ICS, LAMA, and LABA, active components of Anoro (UMEC/VI). sNDA: supplemental new drug application.LAMA : long - acting muscarinic antagonist; COPD: chronic obstructive pulmonary disease; GI: gastrointestinal; JAKi : Janus kinase inhibitor; NRI: norepinephrine reuptake inhibitor. 5 Phase 2 Phase 2 Phase 2b/3 Phase 1 Research Phase 3 Marketed Marketed Filed Research

Economic interest GSK’s TRELEGY ELLIPTA (FF/UMEC/VI): First and only once - daily single inhaler triple therapy

Economic interest in GSK’s TRELEGY ELLIPTA UPWARD - TIERING ROYALTIES OF ~5.5% TO 8.5% OF WORLDWIDE NET SALES 1 Strongest US ELLIPTA launch to date ~31% share in class Marketed in >38 countries, including China launched in 4Q19 sNDA filed 2Q19 for mortality benefit compared with ANORO in COPD sNDA filed 3Q19 for use in asthma 1. TBPH holds 85% economic interest in upward - tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agreement over the next four fiscal quarters). 75% of royalties pledged to service PhaRMA SM notes, 25% of royalties retained by TBPH. All statements concerning TRELGY ELLIPTA based on publicly available information. TRELEGY ELLIPTA is FF/UMEC/VI or fluticasone furoate/ umeclidinium /vilanterol; comprised of ICS, LAMA, and LABA, active components of Anoro (UMEC/VI). COPD: chronic obstructive pulmonary disease; sNDA: supplemental new drug application. 7 20 40 60 80 100 120 140 160 180 200 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 TRx Volume (Thousands) Month Post Launch ANORO ARNUITY BREO INCRUSE Source: GSK, IQVIA NPA weekly TRx data. This information is an estimate derived from the use of information under license from the following IQVIA information service: NPA for the time period Sep 2013 through Nov 2019. IQVIA expressly reserves all rights, including rights of copying, distribution, and republication. TRELEGY Launched in US in November 2017

YUPELRI ® (revefenacin) inhalation solution First and only once - daily, nebulized maintenance medicine for COPD

First and only once - daily, nebulized maintenance medicine for COPD YUPELRI ® (revefenacin) inhalation solution FDA - APPROVED FOR THE MAINTENANCE TREATMENT OF COPD 1. Global Strategy for Diagnosis, Management, and Prevention of COPD, 2018; 2. TBPH market research (N = 160 physicians); ref ers to US COPD patients 3. Loh CH, et al. Ann Am Thorac Soc. 2017 Aug;14:1305 - 11; 4. IMS Health information service: NSP for period MAT May, 2015. Excludes nebulized short - acting beta agonists. IMS expressly re serves all rights, including rights of copying, distribution and republication. LAMA, long - acting muscarinic antagonist; PIFR, peak inspiratory flow rate; LABA, long - acting beta agonist. 9 9 Once - daily LAMAs are first - line therapy for moderate to severe COPD 1 9% of COPD patients (~800,000) use nebulizers for ongoing maintenance therapy; 41% use nebulizers at least occasionally for bronchodilator therapy 2 Nebulized therapy associated with reduced hospital readmissions in low PIFR patients 3
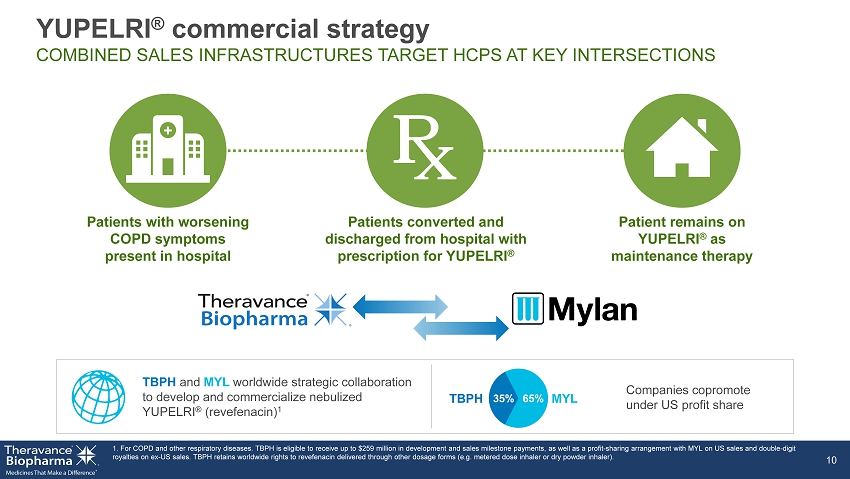
Patient remains on YUPELRI ® as maintenance therapy Patients converted and discharged from hospital with prescription for YUPELRI ® Patients with worsening COPD symptoms present in hospital 1. For COPD and other respiratory diseases. TBPH is eligible to receive up to $259 million in development and sales milestone pa yments, as well as a profit - sharing arrangement with MYL on US sales and double - digit royalties on ex - US sales. TBPH retains worldwide rights to revefenacin delivered through other dosage forms (e.g. metered dose i nhaler or dry powder inhaler). 10 + Companies copromote under US profit share TBPH and MYL worldwide strategic collaboration to develop and commercialize nebulized YUPELRI ® (revefenacin) 1 65% 35% TBPH MYL YUPELRI ® commercial strategy COMBINED SALES INFRASTRUCTURES TARGET HCPS AT KEY INTERSECTIONS

Majority of YUPELRI® volume flows through durable medical equipment channel (approximately 3 month lag in data capture); rema ini ng volume flows through hospitals, retail and long - term care pharmacies. Wholesale acquisition cost (WAC): $1,066 per month (or ~$35 per day). 1. For patients with supplemental insurance. 2. TBPH estimate der ive d from integrating multiple data sources. 3.Effective July 1, 2019. 11 85 wins (equates to 220 accounts) ~70 reviews scheduled (>400 potential accounts) 100% medical support requests fulfilled <30 days FORMULARY Field force productivity goals exceeded ~30,000 patients 2 prescribed (through Q4 2019) PATIENT 100% Medicare Part B 1 ~50% commercial Permanent J - CODE issued 3 ACCESS YUPELRI ® launch metrics STRONG CUSTOMER ACCEPTANCE AND MARKET UPTAKE

Our science Organ - selective approach designed to optimize therapeutic index

Organ - selective approach leverages proven and deep expertise in developing lung - selective medicines for respiratory disease 13 Conventional Systemic Compound Theravance Biopharma Organ - selective Compound Illustrated example: lung selectivity Expanded therapeutic index Narrow therapeutic index EFFECTIVE NON - EFFECTIVE DOSE - LIMITING SAFETY NON - EFFECTIVE DOSE - LIMITING SAFETY EFFECTIVE

TD - 1473 (JNJ - 8398) Oral gut - selective pan - JAK inhibitor Goal: Treat inflammatory intestinal diseases

Improved preclinical profile of a novel, potent, gut - selective pan - JAK inhibitor Tyk2: tyrosine kinase 2. 15 0 20 40 60 80 7.5 8 8.5 9 9.5 10 JAK1 Tyk2 % Absorbed 0 20 40 60 80 7.5 8 8.5 9 9.5 10 JAK1 Tyk2 % Absorbed Percent Inhibitory potency (pKi) Percent Inhibitory potency (pKi) Tofacitinib TD - 1473 (JNJ - 8398) Vehicle treated control group TD - 1473 treatment % Systemic Absorbed % Systemic Absorbed Infiltrating T cells

Systemic exposures low; tissue concentrations at or above JAK inhibition levels Tofacitinib concentrations extracted from J Pharmacol Exp Ther 348:165 – 173, January 2014. BID: twice daily; Hr: hours; IC 50 : concentration to produce 50% maximal inhibition; PK: pharmacokinetics. 16 Plasma Concentrations in UC Patients Colonic Tissue Concentrations of TD - 1473 Tofacitinib 10 mg BID* TD - 1473 20 mg TD - 1473 80 mg TD - 1473 270 mg TD - 1473 Dose Time (h) 1 10 100 1000 10,000 20 mg 80 mg 270 mg Range of JAK IC 50 values 100 200 300 0 0 1 2 3 4 Plasma Concentration (nM) TD - 1473 Total Concentration (nM)

Potential for increased efficacy and safety with gut selectivity 1. Presented at the European Crohn’s and Colitis Organization meeting, March 8, 2019, Copenhagen, Denmark. 2. Component of total Mayo score clinical response. 3. Maintenance phase of the study will have induction responder patients re - randomized to active doses compared to placebo at 44 weeks. 4. Deal value up to $1B in payments to TBPH, including $100M upfront; profit - share in US (33% TBPH, 67% JNJ); double - digit royal ties to TBPH ex - US. 17 44 11 30 20 70 20 73 55 0 20 40 60 80 Rectal Bleeding Improvements Clinical Response Placebo (n=9) TD-1473 20 mg (n=10) TD-1473 80 mg (n=10) TD-1473 270 mg (n=11) TD - 1473 (JNJ - 8398) 1 Phase 1 data in UC; 4 weeks of treatment % of Patients 2

TD - 1473: Gut - selective pan - JAK inhibitor LATE - STAGE STUDIES IN ULCERATIVE COLITIS AND CROHN’S DISEASE Phase 2 Crohn’s and Phase 2b/3 UC studies ongoing Phase 2 Crohn’s and Phase 2b UC data expected late - 2020 Global collaboration with JNJ leverages joint development expertise and provides significant economics to TBPH 4 1. Presented at the European Crohn’s and Colitis Organization meeting, March 8, 2019, Copenhagen, Denmark. 2. Component of total Mayo score clinical response. 3. Maintenance phase of the study will have induction responder patients re - randomized to active doses compared to placebo at 44 weeks. 4. Deal value up to $1B in payments to TBPH, including $100M upfront; profit - share in US (33% TBPH, 67% JNJ); double - digit royal ties to TBPH ex - US. Crohn’s disease Ulcerative colitis Phase 2: 12 weeks (N=160) Dose - finding induction Active treatment extension: 48 weeks Phase 2b/3: 8 weeks (N=240) Dose - finding induction Maintenance phase 3 : 44 weeks Phase 3: 8 weeks (N=640) Dose - confirming induction Responders 18

TD - 8236 Inhaled lung - selective pan - JAK inhibitor Goal: Treat moderate - to - severe asthma regardless of T2 phenotype

High medical and economic burden in uncontrolled asthma 1. 2018 DR/Decision Resources, LLC. All rights reserved. Reproduction, distribution, transmission or publication is prohibited ; reprinted with permission; 2. Sadatsafavi , M., et al. Can Respir J 2010;17:74 - 80; 3. Nurmagambetov T, et al. Ann Am Thorac Soc 2018;15:348 - 56. IFN: interferon; IL: interleukin; STAT: signal transducer and activator of transcription proteins; T2: type 2; TSLP: thymic s tro mal lymphopoietin. 20 Severe 14% Moderate 16% Moderate 25% 16M diagnosed asthma cases 1 Healthcare utilization 2 T2 - high T2 - low IL - 4 IL - 23 /IL - 12 IL - 13 IL - 6 IL - 5 IL - 27 TSLP IFN - γ Bold denotes biologics in development or approved JAK/STAT cytokines implicated in moderate to severe asthma Severe 61 % Inhaled pan - JAK inhibitor has the potential to address patient needs regardless of T2 phenotype Small portion of US patients cause ~$58B in medical costs

-16 -14 -12 -10 -8 -6 -4 -2 0 2 1 2 3 4 5 6 7 TD - 8236: Lung - selective pan - JAK inhibitor PRELIMINARY POSITIVE FENO DATA IN MILD ASTHMATICS FeNO: fractional exhaled nitric oxide. 21 Phase 1 data; mild asthmatics ( 6 - Hour Post - Dose FeNO) Day BL 4000 µg 500 µg 1500 µg 150 µg Placebo - adjusted Change From Baseline (ppb) Phase 1 biomarker study in moderate to severe asthmatics ongoing; data expected mid - 2020

TD - 8236: Lung - selective pan - JAK inhibitor PHASE 2 ALLERGEN CHALLENGE STUDY FeNO: fractional exhaled nitric oxide. 22 Phase 2 allergen challenge study underway Data expected 2020 TD - 8236 Phase 2 Lung Allergen Challenge 12 weeks (N=21) Dose characterization Randomized, double - blind, placebo - controlled, crossover study

Early - stage organ - selective programs

Opportunity to address multiple therapeutic areas with novel organ - selective approach 1. TD - 5202 is being developed in collaboration with Janssen Biotech, Inc. for inflammatory intestinal diseases e.g. celiac disea se. 2. Theravance Biopharma and Pfizer Inc. entered into a global license agreement for skin - selective pan - JAK inhibitors. ALK5i: inhibitor of transforming growth factor β type I receptor. 24 TD - 5202 Oral gut - selective irreversible JAK3i Celiac disease 1 US prevalence: ~3.3 million Intravitreal eye - selective pan - JAKi Diabetic macular edema US prevalence: ~2.7 million Inhaled nebulized lung - selective ALK5i Idiopathic pulmonary fibrosis US prevalence: ~140,000 Inhaled nebulized lung - selective pan - JAKi Prevention of lung transplant rejection ~2,500 per year in US Phase 1 Research Topical skin - selective pan - JAKi Out - licensed: Wholly - owned

Ampreloxetine (TD - 9855) Once - daily norepinephrine reuptake inhibitor for symptomatic neurogenic orthostatic hypotension

Reduced quality of life, significant care - giver burden and limited therapeutic options for symptomatic nOH patients 1. Ha AD, et al. Parkinsonism Relat Disord 2011;17:625 - 8; 2. Mathias C, et al. J Neurol 1999;246:893 - 8. nOH : neurogenic orthostatic hypotension; MSA: multiple system atrophy; PAF: pure autonomic failure; PD: Parkinson’s disease. 26 nOH is a symptom of MSA, PAF and PD ‣ 18% of PD 1 and 83% of MSA 2 patients have nOH ‣ ~350K patients in the US Current treatments have significant limitations ‣ Subset of patients do not respond ‣ None have demonstrated durable effect ‣ Require multiple daily dosing High burden condition ‣ Impact on activities of daily living and quality of life ‣ Significant caregiver burden ‣ Economic burden to the US healthcare system

Untreated nOH NE Release at Neurovascular Junction Systolic Blood Pressure Designed to reduce symptoms of nOH by prolonging the effect of endogenous norepinephrine 1. Palma JA, Kaufmann H. Mov Disord Clin Pract 2017;4:298 - 308. NE: norepinephrine; NET: norepinephrine transporters. 27 Vasodilation Blood pressure Vasoconstriction Blood pressure Syncope Normal x Increase standing blood pressure x Increased brain profusion x Reduce symptoms of symptomatic nOH 1 NE A XON TERMINAL D ENDRITE + Ampreloxetine Ampreloxetine Reduction in syncope Normal

Potential to provide meaningful and durable symptom improvement to underserved patients Baseline OHSA #1 (Orthostatic Hypotension Symptom Assessment Question 1) >4 points. Negative change indicates improvement in symptoms; improvement of 1 point is defined as the MCID (minimal clinically importan t d ifference). ITT: intention - to - treat; SD: standard deviation. Mean (SD) change from baseline in OHSA #1 score n=17 n=13 n=7 n=6 Week Study 169: 4 weeks (N=188) Randomized, double - blind, placebo - controlled, parallel group Study 170: 22 weeks (N=254) Randomized 6 - week withdrawal phase Phase 3 registrational program ongoing; 4 - week efficacy data expected 2H20 28 Phase 3 Registrational Program Ampreloxetine Phase 2 data in nOH; 20 weeks of treatment Extension study: 3 years Completers: -7 -6 -5 -4 -3 -2 -1 0 1 2 3 0 4 8 12 16 20 24 Considered clinically meaningful Withdrawal Durability Efficacy

The Theravance Biopharma Difference

TRELEGY ELLIPTA 1 ‣ FDA approval decision for asthma ‣ FDA approval decision for mortality benefit vs. ANORO in COPD TD - 8236 ‣ Phase 1 Part C data in severe asthmatics ‣ Phase 2 allergen challenge data TD - 5202 ‣ Phase 1 topline data Ampreloxetine ‣ Phase 3 4 - week efficacy data TD - 1473 ‣ Phase 2b/3 ulcerative colitis topline data ‣ Phase 2 Crohn’s topline data Multiple potential milestones and value driving catalysts in 2020 and beyond Commercial progression of YUPELRI ® and TRELEGY ELLIPTA 1. TBPH holds 85% economic interest in upward - tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agreement over the next four fiscal quarters). 75% of royalties received pledged to service PhaRMA SM notes, 25% of royalties received retained by TBPH. All statements concerning TRELGY ELLIPTA based on publicly available information. COPD: chronic obstructive pulmonary disease. 30 2020 2021

Creating transformational value for stakeholders 31 Innovative research yielding organ - selective assets Proven development and commercial expertise Strategic partnerships Value driving catalysts Strong capital position

Strategic objective 32 Transform the treatment of serious diseases through the discovery, development, and commercialization of organ - selective medicines designed to maximize patient benefit while minimizing patient risk

About YUPELRI ® (revefenacin) inhalation solution YUPELRI ® (revefenacin) inhalation solution is a novel once - daily nebulized LAMA approved for the maintenance treatment of COPD in the US . Market research by Theravance Biopharma indicates approximately 9 % of the treated COPD patients in the US use nebulizers for ongoing maintenance therapy . 1 LAMAs are a cornerstone of maintenance therapy for COPD and YUPELRI is positioned as the first once - daily single - agent bronchodilator product for COPD patients who require, or prefer, nebulized therapy . YUPELRI’s stability in both metered dose inhaler and dry powder device formulations suggest that this LAMA could also serve as a foundation for novel handheld combination products . 1. TBPH market research (N=160 physicians); refers to US COPD patients. 33

YUPELRI ® (revefenacin) inhalation solution YUPELRI® inhalation solution is indicated for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD) . Important Safety Information (US) YUPELRI is contraindicated in patients with hypersensitivity to revefenacin or any component of this product . YUPELRI should not be initiated in patients during acutely deteriorating or potentially life - threatening episodes of COPD, or for the relief of acute symptoms, i . e . , as rescue therapy for the treatment of acute episodes of bronchospasm . Acute symptoms should be treated with an inhaled short - acting beta 2 - agonist . As with other inhaled medicines, YUPELRI can produce paradoxical bronchospasm that may be life - threatening . If paradoxical bronchospasm occurs following dosing with YUPELRI, it should be treated immediately with an inhaled, short - acting bronchodilator . YUPELRI should be discontinued immediately and alternative therapy should be instituted . YUPELRI should be used with caution in patients with narrow - angle glaucoma . Patients should be instructed to immediately consult their healthcare provider if they develop any signs and symptoms of acute narrow - angle glaucoma, including eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema . Worsening of urinary retention may occur . Use with caution in patients with prostatic hyperplasia or bladder - neck obstruction and instruct patients to contact a healthcare provider immediately if symptoms occur . Immediate hypersensitivity reactions may occur after administration of YUPELRI . If a reaction occurs, YUPELRI should be stopped at once and alternative treatments considered . The most common adverse reactions occurring in clinical trials at an incidence greater than or equal to 2 % in the YUPELRI group, and higher than placebo, included cough, nasopharyngitis, upper respiratory infection, headache and back pain . Coadministration of anticholinergic medicines or OATP 1 B 1 and OATP 1 B 3 inhibitors with YUPELRI is not recommended . YUPELRI is not recommended in patients with any degree of hepatic impairment . 34
